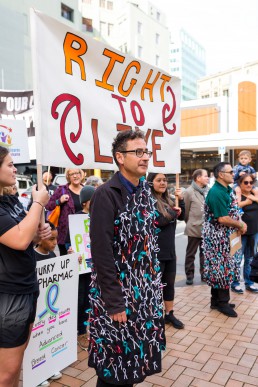Pharmac's dismal ranking little surprise to cancer patients
Pharmac's dismal ranking little surprise to cancer patients
Republished with permission from Graham Adams. Sourced from NOTED.co.nz
By Graham Adams
Aug 16, 2019
New Zealand has the worst record of funding new treatments among 20 OECD countries.
The news this week that New Zealand has been rated the worst among 20 OECD countries for funding modern medicines and pharmaceutical investment overall will come as a surprise to many people but perhaps not to cancer patients.
Cancer treatment gets so much media attention that patients are often acutely aware of what ground-breaking drugs would be available to them if they had the good fortune to live in other developed countries — including Australia. As a result, there is a regular parade of cancer-stricken supplicants to MPs and Parliament begging for their lives and alleging that the funding system overseen by Pharmac is deficient.
The news of our dismal performance came in a report commissioned by Medicines New Zealand, a lobby group for the pharmaceutical industry. It showed that of 304 modern medicines for a range of diseases funded internationally between 2011-17, only 17 were publicly funded in New Zealand.
Australia funded 83, the UK 174, and Germany 178.
And of 70 new cancer drugs funded across the 20 nations in that period, New Zealand had funded only six. Australia’s tally was 26.
Arthritis, asthma, diabetes and cardiovascular disease, among other conditions, fared poorly too.
For all the claims made about Pharmac’s unique approach to funding drugs much more cheaply than in other countries, it is increasingly apparent its strategy of delaying buying drugs until their prices come down is not fit for purpose — particularly when revolutionary cancer treatments are being developed at an astonishing speed.
While Pharmac’s CEO Sarah Fitt is undoubtedly correct in claiming that a “simple count of [funded] medicines won’t tell you how effective a health system is”, she is glossing over the fact that her organisation’s tardy funding decisions amount to rationing by delay for drugs that have proven to be very effective in a battery of clinical trials and medical practice.
As the report noted, the OECD average for approving new treatment from the time of a drug’s registration is 233 days. In New Zealand, it is 512.
As a cancer patient myself, I am very aware of the life-saving drugs available for free in public health systems overseas. I have chronic lymphocytic leukaemia (CLL) and one thing is starkly apparent to me. If my New Zealand-born brother who lives in Brisbane is diagnosed with the same aggressive form of the disease as me, he will be eligible for free access to two wonder drugs — ibrutinib and venetoclax — through Australia’s hospital system. However, New Zealanders with CLL like me can only obtain these drugs through a clinical trial, a drug company’s compassionate access scheme or personally paying more than $130,000 a year (which often means begging from strangers on Givealittle).
Ibrutinib and venetoclax are targeted drugs that have transformed the treatment landscape for those with CLL. Just how revolutionary ibrutinib has been is something Tauranga consultant physician Dr Neil Graham knows only too well from personal experience. Speaking to the Health select committee last week, he had to fight back tears as he recounted how ibrutinib — which he has been able to access through a compassionate access scheme — had saved his life after he had transfusion-dependent bone marrow failure as a consequence of CLL.
He told the committee that ibrutinib has put him into remission and he has been able to continue working productively for the past five years — time that otherwise would have been denied to him, and to the patients he serves.
Writer and TV presenter Clive James is undoubtedly the most famous face of the revolution in CLL drugs. In 2015, he said he was “embarrassed” to still be alive after he had predicted a year earlier that his death — from the double-whammy of chronic lymphocytic leukaemia and emphysema — was likely within months.
Such a prognosis was undoubtedly correct at the time he signalled his imminent demise in 2014 but, soon after that, he began treatment with ibrutinib. His health rallied and he is still alive and writing.
In a book of poems published in 2017 — titled for obvious reasons Injury Time — he described ibrutinib as a “little cluster-bomb of goodness”.
My own experience of coming back from death’s door is similar to that of Dr Graham and Clive James. I knew in 2015 that I was in very serious trouble when a doctor told me rather brutally: “I’ve just read your oncologist’s report. It makes for pretty grim reading, doesn’t it?”
I had been aware that my prospects weren’t very good but the oncologist clearly had been far franker writing to his colleague about my dire prospects than he had been to me in person. I understood the aggressive variant of CLL I have — due to a chromosomal aberration termed “17p deletion” — was difficult to treat but I didn’t realise just how slim the chances of conventional treatment working were.
I had naively assumed there would be something in the oncologist’s armoury that would help. In fact, I learned later that conventional chemotherapy treatment has only a five per cent chance of working for 17p deleted patients. As one haematologist told me: “Standard chemotherapy works well for some 17p patients, but the numbers are very, very small.”
By extreme good luck, I was put in a clinical trial for an experimental drug combination. It held back the progression of my disease for only six months but it made me eligible for another clinical trial of a second-generation version of ibrutinib.
Now, nearly four years later, I am still in remission.
When the drug stops working, if I’m very lucky I’ll get access to a clinical trial for venetoclax and the drug will be provided free. But if I don’t manage to be accepted onto a trial, I’ll have to bankrupt myself to stay alive — as many New Zealand cancer sufferers are doing right now.
However, if I were a citizen of Australia, as my brother is, I would be eligible to have the drug funded for a cost of around $33 a month.
Ibrutinib was first registered by Medsafe in New Zealand in 2015 and subsequently prioritised for funding by Pharmac in 2016.
Venetoclax was registered in 2017 and, last September, Pharmac’s Pharmacology and Therapeutics Advisory Committee recommended venetoclax “be funded with a high priority” for CLL patients with aggressive disease, including 17p patients.
In April this year, the recommendation was repeated as a high priority for funding.
Now, nearly a year later after the recommendation was made last September, the drug remains unfunded.
It is worth noting that Pharmac would undoubtedly be able to buy ibrutinib and venetoclax at a much lower price than its “rack rate”, which could make it not much more expensive per patient than hospital-based dialysis over a year.
Dr Graham wants Pharmac to fund ibrutinib and venetoclax for patients with aggressive CLL disease. He told NOTED: “The timeframes are simply unacceptable for treating malignant diseases generally, and CLL specifically. People are dying waiting for a decision on funding.”
There are about 2000 CLL patients in New Zealand. Dr Graham says most will respond very well to standard chemotherapy and may get remissions of up to 10 years, which is counted as the nearest thing to a cure in treating cancer.
But for the small number who don’t respond to such treatment, their disease quickly becomes a life-or-death matter.
The brutal truth, as he puts it, is that while Pharmac vacillates, people die.
Dr Neil Graham's talk - presented to the Health Select Committee
Dr Neil Graham's talk - presented to the Health Select Committee
The following talk was presented by Dr Neil Graham to the Health Select Committee on 7 August 2019 to support the submission calling for the Select Committee to strongly recommend funding of two life-saving treatments.
“My written submission was to request funding for ibrutinib and venetoclax for CLL, two examples of the new era of cancer treatment medications.
In early 2019 the Cancer at the Crossroads conference was held in Wellington, a major national conference to assess and review the status of cancer treatment in NZ.
An editorial was published in April this year in the NZMJ, reviewing the conference.
To quote from the first paragraph of that editorial:
“Cancer is the leading cause of death in Aotearoa/NZ. The number of those affected by cancer is forecast to increase by 50% in the next fifteen years…..
Our survival rates from cancer lag behind those of Australia, Canada, and Scandinavian countries, and are not improving at the same rate as elsewhere “.
This is a red flag for how we manage the commonest cause of death in our country. We are not doing well by international standards.
In the last decade or so there has been a revolution in cancer medication development internationally, of a magnitude greater, perhaps, than any one area of disease ever. It has been likened in effect to the development of antibiotics for infectious diseases almost a century ago. And it is growing exponentially. It has been predicted that cancer will become controllable, even curable, in the next decade or so.
Some NZers have had their lives saved by these new medications – two of those (Ben and I) sit in front of you today. For me, five years ago, I had transfusion-dependent bone marrow failure. I was likely to die. I was given access to ibrutinib, went into remission, and have been well ever since, working, contributing to society, paying taxes, and living a full life. Ibrutinib saved my life.
Other NZers have not been so lucky.
Most new era life-saving cancer drugs are not funded. If you are wealthy, sell your house to pay, beg in its various guises, move to Australia, or get into a drug trial, you live. If not, you die. And die of a treatable disease. And NZers ARE dying, unnecessarily, because they can’t access these life-saving medications.
Pharmac can’t, or won’t fund them. This is in contrast to Australia, UK, Canada etc, where these medications, including ibrutinib and venetoclax, are funded.
This is still not widely known nationally, but there is an increasing awareness because of this May’s march on Parliament, Blair Vining’s case publicity, and others. When this becomes widely known, it is likely that the NZ population will be critical of NZ health services for allowing this to happen. It is unlikely to be tolerated. You may remember the outcry over hepatitis C in the early 1990’s.
Two other points:
Pharmac has said recently that medications have a relatively small role in cancer treatment. Whilst overall this has some truth, it is not the case for leukaemias, where prevention and screening for early detection have no role, and surgery and radiotherapy have a minimal role. Treatment is all chemotherapy/medications.
And it is the mainstay of treatment in the context of attempted cure in the case of serious cancer in the form of metastatic disease.
Secondly, international guidelines, written by world authorities and specialist societies are not adhered to in NZ, as medications when not funded are generally not able to be used. In CLL, venetoclax, and, even more so, ibrutinib are the first choice medications in international guidelines for severe forms of CLL. But they are not used when not funded.
So people in NZ die unnecessarily.
NZ will increasingly stand out as doing badly in cancer medicine because of this lack of funding – what has been described as third world medicine in a first world country.
So, to finish, the current Pharmac model is not working for modern oncology practice.
My submission makes suggestions regarding this (5 – 9).
The Pharmac model needs to change for modern oncology.
Current Pharmac processing of applications for oncology medications takes almost two years. When you’re dying of cancer, this is woefully inadequate. A rapid access programme for cancer treatments must be developed for these patients.
I would ask/beg this committee to recommend review of Pharmac’s model in modern oncology practice, particularly funding. We spend 5% of our health budget on medications. Other similar countries spend 10 – 15%+.
Involved groups (clinicians, policy makers, Pharmac, patients, and the industry) need to get together and develop a new model to stop the disgrace of NZers dying unnecessarily.”
Health Select Committee hears pleas from survivors and sufferers
Health Select Committee hears pleas from survivors and sufferers
This story was originally sourced here from RNZ
Politicians heard emotional pleas today from cancer survivors, people with disabilities and health agencies to boost Pharmac’s funding for life saving drugs.
Petitions were presented to the Health Select Committee at Parliament – calling on medicines to be funded – including for breast, lung, ovarian cancers, along with multiple myeloma.
Emily Beswick and her daughter, Stella, travelled 500 kilometres from Cambridge to today’s select committee.
Stella has spinal muscular atrophy and uses a wheelchair.
She sat quietly at the back of the select committee room – listening – smiling from time to time.
While medication to help her condition is available overseas – here in New Zealand – it’s not.
And for Ms Beswick that’s devastating.
“We feel that all hope is gone – we have been waiting 13 years for a treatment to arrive and now that it’s finally here – it is out of reach,” she said.
Spinal muscular atrophy is a childhood version of motor neurone disease but the drugs to halt it are not funded here.
MS Beswick said her daughter’s quality of life could be improved.
Neil Graham has been in remission from chronic lymphocytic leukemia for five years.
He broke down as he told the select committee he was given compassionate access to a drug that probably saved his life.
“Since then, I’ve been working, making a useful contribution to society, paying taxes and having a full and enjoyable existence,” he said.
Mr Graham said the Pharmac model needed to change.
“Current Pharmac processing of applications for approval for cancer medications take almost two years – and if you’re dying of cancer – that’s just not acceptable,” he said.
Joy Wilkie is in remission from the blood cancer myeloma.
No new anti-myeloma medicines have been funded here in the last five years.
“When I relapse – and I know I will – our world class clinicians know what best international treatment I need to give me more years of meaningful life,” she said.
Pharmac wasn’t available for an interview and it did not did not appear at the select committee today.
In statement, it said funding decisions take time and it is aware that not everyone has time on their side.
But late this afternoon, it announced on its website a proposal to fund Kadcyla for some breast cancer patients, Alectinib for lung cancer and Ocrelizumab for multiple sclerosis from 1 December.
Meanwhile, a Cancer Action Plan is expected to be announced by Health Minister David Clark later this month.
Leukaemia advocacy group to tell Health Select Committee NZ’s current cancer drug funding scheme is a ‘national disgrace’
Leukaemia advocacy group to tell Health Select Committee NZ’s current cancer drug funding scheme is a ‘national disgrace’
Dr Neil Graham, founder and executive director of CLL Advocates NZ, a new patient support group for New Zealanders with Chronic Lymphocytic Leukaemia (CLL), will appear before the Health Select Committee today in support of their submission to fund life-saving treatments for suffers of CLL.
The Health Select Committee hearing follows the CLL Advocates NZ’s petition, signed by over 1,000 New Zealanders, fighting for funding of ibrutinib (Imbruvica) and venetoclax (Venclexta™).
Neil Graham is calling on the members of the Select Committee to strongly recommend the funding of these two life-saving treatments, to prevent the unnecessary deaths of New Zealanders with the aggressive form of this cancer.
“These treatments are widely funded in many similar countries to NZ, such as Australia, Canada, UK, and Scandinavia, where many lives have been saved. They are not funded in NZ however, and most patients in NZ are unable to afford their cost. As a result, New Zealanders are dying unnecessarily from lack of access to these medications. Those who can pay live, those who can’t die.” said Dr Graham.
Dr Graham is also asking the Select Committee to urgently review the Pharmac model which is patently unable to accommodate the new wave of drugs.
“It is a national disgrace. The current Pharmac model for cancer medication funding is no longer appropriate for providing modern life-saving cancer treatment. It needs to be changed. Funding applications to Pharmac for cancer medications currently take almost two years to be processed. For people dying of cancer, this is woefully too long” said Dr Graham.
Dr Graham, who is living with CLL, had compassionate access to one of these drugs, ibrutinib, and nearly five years later continues to work and live life to the full. He says Pharmac’s continuing refusal to fund the drug is not only a death sentence for New Zealanders with aggressive CLL, but flies in the face of international clinical opinion and best practice.
“Ibrutinib has been registered in NZ since 2015, and is funded in 44 of 45 countries of similar or lower wealth, NZ being the exception. Evidence for the effectiveness of this treatment is incontrovertible, for example overall survival for patients on ibrutinib at first line is 88%. It’s hard to imagine why the evidence that was so compelling for 44 other countries has not persuaded Pharmac to the same position,” said Dr Graham.
Dr Graham will also be joined by Dr Ben Schrader and Dr Rob Weinkove to present in support of this submission.
War in the Blood review – Love, hope and the search for a cancer cure
War in the Blood review – Love, hope and the search for a cancer cure
The feature-length film about a revolutionary treatment for leukaemia did justice to the extraordinary science and the moving stories of the patients involved Martin Pule spent his childhood dismantling radios, computers and any other devices he came across, reassembling them in alternative ways and pressing them into new services. Even allowing for childish curiosity, it speaks to a different way of conceptualising the world – as a miscellany of parts, only temporarily constituted in their current forms. Most of us are just pleased to come across a device already disposed to work.
War in the Blood (BBC Two) was about what happens when such a child grows up, becomes a doctor and turns that mindset towards the human body. “I realised that cells are machines you might take apart, and genetic code is not really that dissimilar to computer programmes.” Now at the University College London’s Cancer Institute, he became a pioneer of CAR T-cell therapy. As he explains it to the viewer, with the unhurried simplicity of someone with knowledge to share and nothing to prove, this is the process of taking a leukaemia patient’s or a donor’s T-cells (the mainstay of our immune systems; they hunt and kill cells infected with viruses) and inserting into them a piece of DNA that teaches them to recognise cancer cells, which they would otherwise ignore, and treat them with the same lethal force they do viruses. Then they return the T-cells to the patient and … well, you hope for the best.
The therapy has recently started phase-one trials – the first tests on humans. The subjects must be terminally ill patients who have run out of treatment options and are willing to undergo the intervention knowing that, of course, it may not work. So far, however, the research suggests that the approach may eventually become a cure for all cancers.
The programme follows two such patients: middle-aged former military man Graham Threader, who was diagnosed with leukaemia in August 2015, and 18-year-old Mahmoud Kayiizi, who has relapsed with the same disease after several rounds of treatment and a bone marrow transplant. Mahmoud looks both younger and much older than his years. “I just want to live,” he says from his hospital bed with a literally bone-deep weariness. “It would be heartbreaking for my mum …” Graham will be treated with his brother’s donated cells, Mahmoud with his own, both weaponised in Dr Pule’s laboratory and administered by a team led by Dr Claire Roddie.
It is a film about love, hope and the tempering of both with realism. Doctors must do it with patients, being careful neither to fan the flames of optimism with their knowledge of the studies coming out of the US (which has more data on the therapy than we do) or douse them with their appreciation of how long any path to any cure must be.
At one point, Mahmoud’s consultant, Dr Ben Carpenter, mentions the risk of something and his mother, Fatuma – her face bright with hope and faith – says: “It’s not a risk. You are saving him.” The doctor pauses, balancing duty with care, kindness with honesty, professional ethics with human impulse. “There are definite risks involved,” he replies gently. “But we know the risks of not doing this are much higher.”
Spouses must guide spouses, as Melly does for Graham, reminding him to weigh his quality of life – and his family’s life – against prolonged suffering in the name of treatment, however pioneering. Sometimes the normal dynamic reverses: patients must guide loved ones and children must do so for parents. After good initial results, Mahmoud cautioned his mother about being “too hasty”. He doesn’t want her to celebrate too early. Much, much older than his years, then.
At an hour and 40 minutes, it was a film that was able to do justice to all its parts. It twined them round each other immaculately, keeping the march of the days and the clinical results clear to the uninitiated viewer without losing sight of the intimacy between everyone involved, or distancing the viewers from Graham or Mahmoud’s experiences, which lay at the heart of everything. It did a particularly fine job of honouring the dual perspective that exists in any clinical trial – the immediate, patient-based focus on individual treatment and outcomes by the administering doctors, and the detached, long-term view the scientists developing the treatment must take – without letting the more naturally sympathetic former outweigh or diminish the latter.
There was agony and ecstasy by turns, and endless courage from Graham and Mahmoud and the people that love them. Lives and cells were taken apart and put back together in different forms. Godspeed, everyone.
March for the Right to Live
March for the Right to Live
On Thursday 7 May hundreds of cancer sufferers marched to the steps of Parliament to ask that the drugs that extend or improve the quality of lives are funded by Pharmac.
Watch the video of the march and Ben Schrader’s speech below.
In praise of big pharma: The Indefensible position
In praise of big pharma:
The Indefensible position

I know, I know, Big Pharma are bastards! They rig clinical trials, bribe doctors to prescribe their products, medicalise everyday problems by casting them as diseases that suddenly “need” expensive medication to control them and spend much more on marketing than they ever do on original research. And that’s before we take into account tax dodges and the shameful role pharmaceutical companies play in scandals such as the opioid epidemic in the US. They are up there with Big Oil and Big Banks in the array of hellish multinationals we all love to hate.
Except I can’t really join the jeering throng myself. I literally can’t live without Big Pharma’s products. No amount of echinacea or rescue remedy or bark carefully collected from trees in Vietnam’s highlands is going to reverse or even hold the progress of my chronic lymphocytic leukaemia.
I know people who believe their “natural” potions are what keep them healthy and that they will be effective against serious illness when it strikes but they seem to change their minds when their health actually collapses. They are suddenly happy to sidle off to conventional doctors to get the treatments they once claimed to be the work of the devil – aka Big Pharma.
No amount of rescue remedy is going to reverse or even hold the progress of my chronic lymphocytic leukaemia.
One acquaintance, who enthusiastically endorses complementary medicines and nutritional supplements, was clearly disapproving when she heard I had enrolled in a clinical trial for a new targeted cancer drug. She scowled and said: “Oh, so you’re going down the chemical route, are you?”
Sure was! And gladly! What’s more, she was too when she later developed a chronic, debilitating disease and found that only Big Pharma’s products could help turn her into a functioning human being again.
If there are no atheists in a foxhole, there are also very few cancer patients whose first port of call is a homeopath. Such people do, of course, exist, but not for very long.
Big Pharma may be bastards but at least their products mostly work, at least to some degree, and often astonishingly well. In my case, they have given me at least another two years of life so far and the hope of more to come.
Of course, Big Pharma should be criticised and held to account for their shadier practices – and, god knows, there are plenty of them. But I’d hate to live in a world without their products. Thanks to treatments made widely available by Big Pharma, tuberculosis, polio, Aids and other diseases that once killed or disabled thousands upon thousands can be controlled.
I know someone who had hepatitis C for decades after a misspent youth. A year ago, he – a burly builder – wept when he was told the revolutionary drug he had been prescribed had cleared the virus from his body. His crippling fatigue vanished and his productive life has been restored.
Serious childhood infections such as measles, diphtheria, rubella, mumps and tetanus have been mostly wiped out by vaccines. In my lifetime, the number of deaths from coronary heart disease has been slashed and life expectancy for many cancer patients keeps increasing.
The most widespread criticism of Big Pharma centres on the cost of their drugs. The experimental drug I am taking is yet to be approved for chronic lymphocytic leukaemia by the US Food and Drug Administration, but it is likely it will cost at least $10,000 a month when it is commercially available. And this is a drug that is not a cure and which patients need to take until the wily cancer finds a way around its defences.
When that happens, I may have to start paying through the nose for another drug, but if I’m really, really lucky I’ll be accepted onto another clinical trial.
The day I was told I had been accepted onto my first clinical trial in 2015, I was so happy I tipped the waitress who brought me a hot chocolate in the hospital cafeteria a $20 note. She was taken aback but I told her: “I’ve had a very lucky day!”
In fact, those drugs didn’t work very well and put me in A&E a few times but they cleared the path for me to get onto a second trial. After three years of treatment, my blood is finally normal for the first time since 2013. And not only are the drugs free but the pharmaceutical company gives me a $10 petrol voucher and pays for my parking each time I visit the hospital, which is a nice touch.
I have swallowed at least $360,000 worth of the company’s drugs so far on this trial and if I’d had to pay for them myself, I’d have had to sell my house by now and be facing eventual bankruptcy just to stay alive.
It seems entirely wrong that a company would charge so much for its life-saving products until you consider how long and hard the road to a successful drug is. It can take a decade or more to progress from laboratory to market. The cost of developing a new drug has been estimated as somewhere around $US160 million (by consumer advocates) to $US2.6 billion (by the pharmaceutical industry) but if the lower figure is anywhere near correct you’d imagine there would be many more entrants into the market given the money to be made in such a profitable business.
The best justification I can make for paying high drug prices – and the way I’ll console myself if I have to start paying exorbitant sums to stay alive – is to imagine being up on a murder charge. Whether I was guilty or innocent, I’d still want the best lawyer in town to keep me out of jail and give me my life back, even knowing he or she would charge like a wounded bull. I certainly
wouldn’t be haggling over the price to save my skin.
If that cost me hundreds of thousands of dollars, I’d probably consider the eye-watering sum worth it if I could raise it. I wouldn’t, of course, be thrilled to pay the bill – especially if I ended up in jail for a long stretch despite my counsel’s efforts – but I’d always take the chance, even if I was bankrupt at the end of it.
Paying for life-saving drugs that give you another shot at living is like that, even though – like an expensive defence lawyer – it’s always possible they may not succeed.
Governments and public bodies are always welcome, of course, to take the million- or billion-dollar risks that Big Pharma routinely does in developing new drugs. But it’s hard to imagine the public would have any appetite to be told on Budget day: “The government would have been in surplus without the $1 billion loss on our state-funded experimental drug development programme after all our clinical trials failed. We will have to make spending cuts all round.”
And guess who we are all going to turn to when increasing antibiotic resistance goes from being confined to relatively isolated incidents and suddenly becomes a widespread problem – particularly for the very young and the very frail? Not to mention when surgery for even routine procedures such as hip replacements and chemotherapy for cancer become extremely dangerous?
Already scientists fear losing control of malaria and TB and sexually-transmitted infections such as gonorrhoea, syphilis and chlamydia as antibiotic resistance grows. Faced with that possibility, the UK government has turned its mind to providing incentives for Big Pharma to develop new antibiotics.
Big Pharma prefers to create blockbusters that earn $US1 billion or more. The easiest paths to profitability are drugs to treat chronic conditions such as heart disease, not antibiotics that are taken as a one-off course for an infection. So the British government is mulling setting up a $US1 billion prize to encourage big pharmaceutical companies to develop new and effective antibiotics.
Although drug companies are seen – with good reason – as rapacious, they do have a softer, more generous side: Big Pharma gives away the greatest proportion of its profits of any industry. In 2013, biopharmaceutical companies led all other corporations by donating 19.4% of their profits before tax to charitable groups.
Most of this beneficence is in-kind product – ie, drugs – that also drums up new business and generates tax benefits, but it’s a gift nevertheless. The good news is that even if you’re not eligible for a clinical trial, you may get compassionate access to a drug that could transform your life. Big Pharma may be bad – perhaps very bad – but they’re not all bad by any means.