Pharmac decision to fund two treatment combinations and widen access to ibrutinib for CLL
We are very pleased to announce that Pharmac has announced new funded treatment options for people with chronic lymphocytic leukaemia (CLL).
From 1 May 2026, two fixed‑duration combination therapies, venetoclax + ibrutinib and venetoclax + obinutuzumab, will be funded as first‑line treatments.
Access to ibrutinib monotherapy is also being widened so it can be used as a second‑line option for anyone whose CLL has relapsed or caused intolerable side effects.
These changes will give more people access to modern, effective treatments and reduce pressure on hospital infusion services.
Pharmac has also updated criteria so people currently self‑funding can transition to funded treatment within six months.
❤️ We’re grateful to our members who have shared their stories, your voices, alongside the work of the CLLANZ Trust, have helped influence better options and outcomes.
CLL Advocates Pharmac Submission
CLL Advocates NZ has made a formal submission to Pharmac supporting the proposal to fund two time‑limited treatment combinations for first‑line CLL care: Venetoclax with Ibrutinib, and Venetoclax with Obinutuzumab.
This represents an important step toward giving New Zealanders access to modern, internationally recognised therapies. Our submission welcomes the proposal, outlines several refinements to ensure equitable access for patients who have been self‑funding treatment, and highlights the need for flexibility where side‑effects or clinical circumstances require alternative options.
Here is a link to the submission: CLL Pharmac Submission
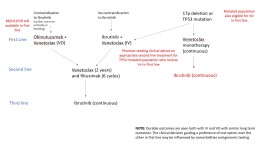
Here a link to the flowchart for the Proposed Funded CLL Treatment Pathway: Treatment Pathways
CLL Advocates March Newsletter
CLLANZ News
Since our last report to members your Trustees have been busy.
We attended our usual online briefings with Pharmac and the Blood Cancer Alliance and also spoke with Blood Cancer NZ to provide feedback on a State of Blood Cancer Report, which is scheduled to be released in Parliament this April.
More on these items later.
Ibrutinib + Venetoclax
Our biggest news in recent times has been the announcement by Pharmac that I+V will be a funded frontline treatment for treatment-naïve CLL patients with the expectation being that, on average, 80-90 people will benefit from this every year. This signals the end of chemo-immunotherapy (FCR) for all newly diagnosed patients.
It has, however, caused some anxiety among those CLL patients who have already been treated at least once. It is not yet clear what treatment options will open up for these people, and we will be seeking clarification from Pharmac.
We managed to get some good media coverage at the time.
We will also continue lobbying vigorously for treatments not yet available in NZ but available and funded in many other countries.
State of Blood Cancer Report
This is a comprehensive document which will be a useful source of information for some time to come once it is published. BCNZ is going through the final pre-production process now.
The Report will be a useful lead into the Election.
Election Campaign
A collection of Cancer NGOs plus organisations such as Myeloma NZ and CLLANZ is funding what will be a very slick campaign aiming to draw the public’s attention to the bareness of NZ’s medicine cupboard. We will provide further updates as the process takes shape.
Trustee News
Ruth Spearing, Haematologist, Christchurch has returned to the role of Chair following the sad death of her husband in early January. We sent a tribute to Ruth on behalf of you all.
Lisa Ryan, CLL patient, Auckland has taken the role of Deputy Chair.
CLLANZ Website
We’re working towards refreshing the CLLANZ website to improve usability and presentation, and we have funding applications underway to help make this possible. If you have skills in web design or development and would like to contribute, we’d be grateful to connect with you.
Global Survey
The Lymphoma Coalition has put together an international survey for people with lymphoma or CLL. This will help create information as to how New Zealand is doing compared to other countries. Where a country has more than 100 responses, the Coalition will collate the country's data and send it back - in our case to Blood Cancer New Zealand, who we work very closely with on all sorts of issues. For the data to be meaningful - and therefore usable - for CLL, it would be great to have as many people as possible with CLL to respond to the survey.
Here is a link to the survey: lymphomacoalition.org/global-patient-survey/
Please note that the survey closes on 31 March 2026
It takes 20- 30 minutes to do the survey, depending on how many treatments you have had, so make yourself comfortable, make yourself a coffee, and thank you for filling it out!
Please feel free to pass it on to other people with CLL - or any form of lymphoma.
Data help us advocate more successfully so thank you for doing this!
Rob Crozier
On behalf of the Trustees CLL Advocates NZ
Proposed drug funding for blood cancer welcome, but NZ still lagging behind
PRESS RELEASE - Featuring Marc Pearce, CLL Advocates Trustee
Proposed drug funding for blood cancer welcome, but NZ still lagging behind
Joanne Naish [thepress.co.nz]
February 18, 2026
Nelson man, Marc Pearce, is living with CLL and welcomes Pharmac's proposal to fund new drugs.
A proposal to fund new drugs for one of the most common blood cancers is welcome news, but patients and advocates say New Zealand still lags behind other countries.
Pharmac is consulting on whether it should fund two new treatment combinations - venetoclax with ibrutinib, and venetoclax with obinutuzumab - for people with chronic lymphocytic leukaemia (CLL). If approved, funding would begin on May 1 this year.
Nelson man Marc Pearce was diagnosed with incurable CLL in October 2024 after a routine blood test, but does not need treatment yet.
He said the announcement was a huge weight off his mind, as well as all the people awaiting or having other outdated treatments which had unpleasant side effects and had been abandoned in other countries like Australia, Canada and the UK.
Patients and advocates say access to modern medicines is constrained by Pharmac's budget.
He said many people were self-funding the drugs at a cost of about $1500 per month or going overseas for treatment.
But while he welcomed the announcement, access to new medicines in New Zealand remained well behind other countries in the OECD with a similar GDP per capita.
“We're the worst country in all of the countries in the OECD in terms of getting access to those new medicines. So whilst this gives us some access, it doesn't solve the problem,” he said.
He said it was estimated that Pharmac would need to spend $6 billion a year instead of $1.8 billion a year to get up to the average per capita funding within the OECD.
“So, whilst this is great news for the 3000 people in New Zealand or so who have got CLL, it doesn't fix the overall long term structural problem in terms of funding medicines.”
CLL Advocates NZ chairperson Ruth Spearing said it was an exciting announcement for the organisation which had been campaigning hard to get these treatments funded.
“We welcome the fact that these treatments are being offered as frontline therapy to patients with CLL and that patients will no longer have to have had old fashion, toxic and indeed cancer-causing chemotherapy and then relapsed before becoming eligible for more modern treatments,” she said.
The new treatments are in oral pill form and do not require IVs or hospital stays like traditional treatments like chemotherapy.
The oral treatments would free up hospital time and allow patients to continue their normal lives while undergoing treatment, as well as extend their lives.
“The funding of drugs in this country still remains at the bottom of all the OECD countries despite our GDP per capita being in the middle our OECD partners,” she said.
“We call on this government and the subsequent governments to rectify this situation so more lifesaving drugs can be funded for CLL and other haematologic conditions.”
Pharmac’s Director Pharmaceuticals, Adrienne Martin said the proposal would give around 80 to 90 people each year access to powerful, targeted treatments immediately after their diagnosis.
“We’ve heard clearly from the blood cancer community and from our clinical advisers that these combinations could make a real difference when used early,” she said.
CLL is a slow‑growing blood cancer where the bone marrow makes too many abnormal white blood cells. While it cannot be cured, people can live well for long periods with the right treatment.
Pharmac is seeking feedback by March 4.
Associate Health Minister David Seymour said improving access to cancer medication in New Zealand was important to cancer patients, and their families.
Associate Health Minister David Seymour says while CLL is not curable, the new drugs would extend people’s lives.
“While CLL isn’t curable, the right treatment means patients can live longer more fulfilling lives. Venetoclax with ibrutinib or obinutuzumab will help patients achieve longer lasting remission, and avoid the need for traditional chemotherapy. Two of these medicines will be available in pill form, which don’t require an IV drip and could mean fewer hospital visits,” he said.
He said people currently paying privately for these combinations could receive funded treatment in a private hospital, provided they met the funding criteria at the time they began treatment to avoid the hugely disruptive and stressful transfer to a public hospital during an immensely difficult period.
Health Minister Simeon Brown said the Government was committed to improving treatment and care for New Zealanders with cancer, including blood cancer.
“That’s why this Government has funded seven new cancer medicines that target blood cancer through Pharmac’s $604 million investment – expanding treatment options for patients and their families.”
It was also significantly expanding stem cell transplant services for patients with blood cancers and related conditions, helping more people access critical, life-saving treatment sooner, he said.
Media release- Funding of Chronic Lymphocytic Leukaemia with Ibrutinib + Venetoclax and Obinutuzumab + Venetoclax
Chronic Lymphocytic Leukaemia Advocates NZ (CLL Advocates NZ) welcomes the announcement by Pharmac that the combination therapies of Ibrutinib + Venetoclax and Obinutuzumab + Venetoclax will be funded for Chronic Lymphocytic Leukaemia (CLL) patients by haematologists as initial treatments. CLLANZ has been campaigning hard to get these treatments funded.
“We welcome the fact that these treatments are being offered as frontline therapy to patients with CLL and that patients will no longer have to have had old fashion, toxic and indeed cancer-causing chemotherapy and then relapsed before becoming eligible for more modern treatments.” said Dr Spearing, Chair of CLL Advocates NZ.
“These oral treatments will free up hospital time and allow patients to continue their normal lives while undergoing treatment. It will also extend lives in a meaningful way,”
“Having both combinations of ibrutinib and venetoclax and obinutuzumab with venetoclax funded will now enable haematologists to deliver much safer and effective CLL treatment as first-line treatment to patients at all ages, which is especially exciting.”
“Although it is great news that these drugs have at last been funded - and we appreciate the efforts of all those who have made this possible - the reality is that we are well behind other countries with a similar GDP per capita. The funding of drugs in this country still remains at the bottom of all the OECD countries despite our GDP per capita being in the middle our OECD partners. We call on this government and the subsequent governments to rectify this situation so more lifesaving drugs can be funded for CLL and other haematologic conditions.”
“CLL Advocates NZ will continue to fight for better treatments for CLL patients. CLL is one of the most common blood cancers, with an increasing incidence in the aging population.”
CLL Advocates Trustees
Season’s Greetings – December 2025

🎄 A Message from Your Trustees - Season’s Greetings to all CLL patients, your caregivers, and whānau.
🌟 Trustee Updates
CLL Advocates NZ is becoming more active as we look ahead to a General Election less than a year away.
1. New Trustees:
We welcome Bronny Colquhoun, Craig Marshall, and Sue Osborne – all New Zealanders living with CLL who bring valuable skills to our mission.
2. Patient Support Officer:
We are pleased to appoint Ingrid Ovenstone, whose husband was recently diagnosed with CLL.
Learn more about our team on the CLL Advocates About Us page
3. We continue to benefit from the expertise of Amy Holmes, Consultant Haematologist at St George’s Cancer Care Centre in Christchurch, as our Medical Director.
4. We are also privileged to have Ruth Spearing, a distinguished New Zealand haematologist and global leader in clinical trials, as part of our advisory team.
🗣 Opinion Pieces
With election year approaching, our focus is on advocacy and patient voices.
1. Ingrid Ovenstone has written an opinion piece – read it here
2. Bronny Colquhoun’s piece is in progress and will be published soon.
3. Together with Blood Cancer NZ (formerly Leukaemia & Blood Foundation), we are contributing to a major election-year campaign for better treatment options and outcomes.
💊 Pharmac News
Rob Crozier has been participating in monthly Consumer Stakeholder meetings with Pharmac executives.
Key updates from the 10 December meeting:
1. OFI Consultation: Pharmac has decided not to proceed as proposed. They are considering alternative options and will make a formal announcement before Christmas.
2. 2026 Budget Bid: Pharmac is one of the few Government entities allowed to bid for extra funds. Most others face freezes or cuts.
🌐 CLLANZ Website Refresh
We are updating and refreshing our website to make it more useful for patients and caregivers.
If you have suggestions, please email us at info@clladvocates.org.nz.
Visit us anytime at clladvocates.nz
💌 Closing Note
Till next time… stay well.
Warm regards,
Rob Crozier
On behalf of the Trustees CLL Advocates NZ
Pharmac Proposal Risks Cutting Access to Medicines
Ingrid Ovenstone, Patient Support Officer with Chronic Lymphocytic Leukaemia Advocates NZ, warns that Pharmac’s plan to remove medicines from its Options for Investment list after two years hides chronic underfunding.
These medicines have already been proven effective and cost‑efficient, but New Zealand’s low spend on medicines - just 4.9% of the health budget compared with an OECD average of 13.3%, leaves patients waiting years for treatments that are standard overseas.
For people with conditions like chronic lymphocytic leukaemia, this means relying on outdated chemotherapies while modern oral and targeted therapies remain unfunded.
Read the article here: Pharmac Proposal Risks Cutting Access to Medicines
CLLANZ Submission on Proposed Changes to the Options for Investment List
Chronic Lymphocytic Leukaemia Advocates of New Zealand (CLLANZ) has submitted formal feedback on Pharmac’s proposal to decline a number of medical funding applications currently listed on the Options for Investment (OFI) list. Representing the interests of CLL patients, their caregivers, and specialist haematologists, our submission raises serious concerns about the potential removal of medicines that have been clinically recommended but remain unfunded.
While we acknowledge the realities of prioritisation within the funding system, we strongly oppose the blanket removal of lower-ranked medicines without transparent consultation. Our submission calls for a more robust and inclusive process - one that involves clinical sub-committees and patient advocacy groups before any decisions are made.
We invite you to read our full submission below:
"On behalf of Chronic Lymphocytic Leukaemia Advocates of New Zealand (CLLANZ) we are submitting on the proposal to decline some medical funding applications on the Options for Investment List (OFI). We do so on behalf of CLL patients, their caregivers, and haematologists with a specialist interest in this field.
Whilst we acknowledge that there will always be some aspect of “prioritising” and “rationing” when it comes to deciding which medicines to fund, we are completely opposed to the idea that a significant number of medicines currently ranked near the bottom of the list should be eliminated arbitrarily and en masse as proposed.
Drugs are placed on the OFI after being highly recommended by clinical sub-committees. We recognise that, in some cases, a particular medicine may be superseded by an improved product. However, removal of any drug from the OFI should involve these two crucial steps:
- discussions with the clinical sub-committee that made the initial recommendation; and
- a wider phase of consultation with patient and consumer groups including publication of the discussions with the clinical sub-committees.
This approach will only be successful, however, if Pharmac is more transparent with information regarding the drugs that are in these lower rankings, how long they have been on the list and what their exact rankings are.
There may be valid reasons why a medicine should remain on the list and it is essential that any group with a particular interest in a drug is given the opportunity to express its views on the matter before any decisions are made. This may even open up the possibility that a drug company would be willing to bundle in a low-ranked drug as part of a package of drugs under negotiation.
We would be pleased to discuss any aspect of this submission with you."
Ruth Spearing Rob Crozier
Chair Trustee
CLLANZ CLLANZ
"Cancer patients have not been forgotten - Luxon" (few blood cancer drugs funded)
Prime Minister Christopher Luxon says he has not forgotten blood cancer patients but will give no assurance that a campaign commitment will be fulfilled.
Luxon promised that blood cancer patients would not be forgotten, something many suffering from the disease have called a 'broken promise'.
"I appreciate there'll be classes and categories of people that are still suffering and that would still love a range of medicines on a range of health issues across New Zealand, that's why we're working incredibly hard to grow our economy so we've got even more funding, more money going in," he said in response to questions from Checkpoint.
"I'm sure that there's more we can and will do."
National promised to close the medicine gap and Luxon said his government had delivered more than any other in recent history.
"We're delivering, we put $605 million into cancer drugs we've got 66 new cancer medicines or medicines that we didn't have before we made that investment. We've got six blood cancer drugs as I see it as part of that."
Leukaemia & Blood Cancer New Zealand said the six cancer blood cancer drugs the government had funded were only applicable to less than 1 percent of people suffering from the disease, and none of the six medicines were for treatment of myeloma.
Some myeloma patients have had to move to Australia to access treatment, while others have taken out their Kiwisaver to fund a life-prolonging medicine called daratumumab.
During the election campaign, National pledged to fund a list of medications for solid cancer tumours, and it was forced to make good on that following public backlash over a lack of action.
The prime minister remained resolute that since following through on that commitment, the government had delivered for cancer patients, but it wasn't up to him for when more would be delivered.
"Those are decisions for health and Pharmac and they'll continue to do that job.
"But I think we should be very proud as I talked to cancer victims up and down this country who have actually been really helped and had pain and suffering and life extended because of the commitment that this government made, that the previous government didn't make."
Luxon did not say whether the government would be funding further treatments for blood cancer patients.
"Go talk to the people that are now getting Keytruda funded through this government and through Pharmac and the life changing differences that that's made to many people up and down this country."
Click the link below to read the full article
https://www.rnz.co.nz/news/national/574268/cancer-patients-have-not-been-forgotten-luxon
Blood Cancer Patients Urge Government to Honour Drug Funding Promise
In this RNZ Checkpoint segment, blood cancer patients, clinicians, and advocates speak out about the urgent need for the New Zealand Government to fund critical medications for blood cancers, many of which remain unfunded despite pre-election commitments.
Unfulfilled Commitment: The National Party pledged to fund cancer treatments for both solid tumours and blood cancers. While solid tumour drugs received funding after public pressure, blood cancer medications were excluded from Budget 2025.
Clinical Voices:
- Dr. Rodger Tiedemann, consultant haematologist at Auckland Hospital, describes the lack of funding as a “fundamental health policy failure.”
- He and 59 haematologists co-signed a letter urging the government to act, emphasizing that blood cancer patients rely solely on medication, unlike solid tumour patients who may benefit from surgery or radiation.
Patient Impact:
- Patients with chronic lymphocytic leukaemia (CLL), lymphoma, and other blood cancers are left waiting for access to treatments that are funded in dozens of other countries.
- Many are paying privately or going without, while Pharmac’s “options for investment” list continues to grow without action.
Advocacy in Action:
- CLL Advocates NZ and other groups are calling for transparency, urgency, and equity in cancer drug funding.
- The segment underscores the emotional and financial toll on families, and the need for government accountability.
🎧 Listen to the Full Segment here