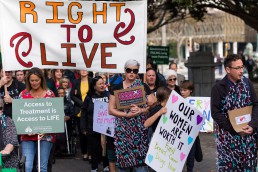Season’s Greetings – December 2025

🎄 A Message from Your Trustees - Season’s Greetings to all CLL patients, your caregivers, and whānau.
🌟 Trustee Updates
CLL Advocates NZ is becoming more active as we look ahead to a General Election less than a year away.
1. New Trustees:
We welcome Bronny Colquhoun, Craig Marshall, and Sue Osborne – all New Zealanders living with CLL who bring valuable skills to our mission.
2. Patient Support Officer:
We are pleased to appoint Ingrid Ovenstone, whose husband was recently diagnosed with CLL.
Learn more about our team on the CLL Advocates About Us page
3. We continue to benefit from the expertise of Amy Holmes, Consultant Haematologist at St George’s Cancer Care Centre in Christchurch, as our Medical Director.
4. We are also privileged to have Ruth Spearing, a distinguished New Zealand haematologist and global leader in clinical trials, as part of our advisory team.
🗣 Opinion Pieces
With election year approaching, our focus is on advocacy and patient voices.
1. Ingrid Ovenstone has written an opinion piece – read it here
2. Bronny Colquhoun’s piece is in progress and will be published soon.
3. Together with Blood Cancer NZ (formerly Leukaemia & Blood Foundation), we are contributing to a major election-year campaign for better treatment options and outcomes.
💊 Pharmac News
Rob Crozier has been participating in monthly Consumer Stakeholder meetings with Pharmac executives.
Key updates from the 10 December meeting:
1. OFI Consultation: Pharmac has decided not to proceed as proposed. They are considering alternative options and will make a formal announcement before Christmas.
2. 2026 Budget Bid: Pharmac is one of the few Government entities allowed to bid for extra funds. Most others face freezes or cuts.
🌐 CLLANZ Website Refresh
We are updating and refreshing our website to make it more useful for patients and caregivers.
If you have suggestions, please email us at info@clladvocates.org.nz.
Visit us anytime at clladvocates.nz
💌 Closing Note
Till next time… stay well.
Warm regards,
Rob Crozier
On behalf of the Trustees CLL Advocates NZ
CLL Advocates August Newsletter
CLL Advocates August Newsletter
Greetings
Your Trustees have been active since we sent out our last newsletter.
Catherine Isaac, Chair, has resigned and we wish to pay tribute to her:
Thank You, Catherine Isaac
We extend our heartfelt thanks to Catherine Isaac for her dedicated service to CLL Advocates NZ since its inception. Catherine has recently retired from her role as Chair.
Her commitment, insight, and leadership have made a lasting impact on our work and the lives of those affected by CLL. From championing access to life-changing treatments to advocating for systemic change in the Pharmac model, Catherine has been a steadfast advocate and a source of wisdom and encouragement for the team. Catherine’s contributions have left a lasting legacy and we are grateful for the time and energy she has devoted to our cause.
Thank you, Catherine, for everything you’ve done.
Trustees update
Ruth Spearing (CLL Advocates Trustee) has been acting as de facto Chair and keeping us all busy! Ruth has now been elected as Chair.
Rob Crozier made a submission on the Medicines Amendment Bill in May, a copy of which is available on our website: www.clladvocates.nz
Rob was also present at the reading of the budget in Parliament at the invitation of Malcolm Mulholland (Patient Advocate Voice Aotearoa) and managed to make the case about the lack of funding for blood cancer medicines to Radio NZ. The media release has been posted on our website.
Rob has also taken on the role of participating in monthly online meetings of the Pharmac Consumer Advisory Committee which is attended by the Pharmac CEO and senior executives. Pharmac has recently established a Patient Advocacy Network chaired by Malcolm Mulholland with Tim Edmonds, CEO of Leukaemia and Blood Cancer NZ as a member.
The current key concern for us (and for haematologists) is getting Pharmac to approve the use of Venetoclax and Ibrutinib in combination. We are waiting for an update as to where it sits on the priority list. Tim Edmonds raised the need for a Modern Medicines Access Strategy — an idea that we should get behind.
Marc Pearce (Trustee) has taken on responsibility for keeping the clinical trials information on our website and Facebook page accurate and has been researching comparative international data on the proportion of Budget: Health being spent on new medicines.
Lisa Ryan (Trustee) is taking responsibility for our website, alongside Melanie Murray, our valued administrator.
TRUSTEE’S MEETING 6 August
Trustees met in an online meeting and began with a lengthy presentation from Matt Tyson, Janssen, reporting on the stalled approval process for Ibrutinib + Venetoclax for CLL. We had a long discussion about strategy for moving this forward and are now working on implementing that strategy. If you are currently self-funding this treatment or either of the drugs separately we’d like to hear from you.
Trustee vacancy
Trustees decided to defer discussion re adding an additional Trustee to the next meeting — scheduled for 8 September. If you’re reading this and feel you have something to offer watch this space...
Declaration calling for urgent government action
New Zealand’s health system is in real trouble, and a group of passionate health organisations is asking Kiwis to help turn things around.
They’re inviting everyone to sign a Declaration calling for urgent government action, and you can add your name at one of the 16 special events near you on Tuesday, 18 November 2025.
The campaign is backed by Patient Voice Aotearoa, NZNO, RNZCGP, BHAG, Hauora Taiwhenua, and ASMS. Keen to help?
Below is a flyer with information on the Wellington event.
For the other 15 locations, please head to https://www.patientvoice.nz/petition-locations to learn more.
CLL Advocates Newsletter May 2025
Greetings
We are very pleased to let you know that CLL Advocates NZ is up and running again, reset and refreshed with new trustees and a sharp focus on our mission. After losing three of our five founding trustees it’s taken us some time to rebuild, but we’re pleased to introduce and welcome our three new trustees, Rob Crozier, Marc Pearce and Lisa Ryan, all New Zealanders living with CLL and with great skills to bring to our mission. Read about them here on our ‘About us’ page.
We’re also very fortunate to have Amy Holmes, Consultant Haematologist at St George’s Cancer Care Centre in Christchurch, as our Medical Director, in addition to the very valuable support of Ruth Spearing, a highly distinguished New Zealand haematologist and leading light in the world of clinical trials.
Our thanks for their contributions go to founding Trustee Dr Gillian Corbett, and Trustee Diane Ward who retired from their respective roles as Medical Adviser and Trustee last year.
Strategy workshop
Our board met last month in Wellington in person and with two via video link. Discussions centred round the future direction of CLLANZ, maintaining our focus, strengthening our alliances, increasing the number of people we reach and what our immediate activities should be.
Our meeting included two very helpful in-person sessions:
Dr Rob Weinkove, one of NZ’s leading CLL experts and a pioneer in CAR-T cell treatment, spoke on current and future CLL management in New Zealand and what we can do to help, and Todd Stephenson, ACT MP, who works closely with David Seymour on Pharmac, Medsafe and related health matters, updated us on ongoing Pharmac changes and opportunities to advocate for improvements in medicines access.
See a summary of Rob’s talk here
As a result of this latter discussion, we are preparing a submission in support of the Medicines Amendment Bill which proposes to speed up the process whereby Medsafe approves medicines for use in New Zealand if they have been approved by two recognised overseas jurisdictions. The bill would also enable more flexible approaches to the prescription and administration of medicines so that patients can get access to the medicines they need more efficiently. Submissions are due on 19 May.
Clinical trials
Our page on clinical trials is now being updated regularly, most recently on 15 April, so keep an eye on these if this is of interest to you.
Participation
Our organisation would benefit greatly from an influx of new people either by joining our Facebook page or by registering on our website. Although our Facebook group membership has doubled in the last two years (to 227) we are still only reaching about 10% of New Zealanders living with CLL. It’s important for us to get more people involved, be they patients or caregivers, as that adds weight to our voice when we’re lobbying for access to new treatments etc. Word of mouth is probably our most effective way of getting the message across. If you know of others with CLL please advise them of either or both of these options - our website and our CLL Facebook page and group.
Finally, we’re in the process of refreshing and updating our website, so if you have any ideas on how we can make this more useful please do get in touch at: info@clladvocates.org.nz
Till next time…. Stay well.
Catherine Isaac
On behalf of the Trustees
CLL Advocates NZ
CLL Advocates June Newsletter
Dear Friends of CLL Advocates
We have a lot to report on and a lot to get on with!
CLL Focus groups
We’re pleased to share an overview of key findings from the CLL patient focus group discussions we held in December 2022 (Focus Groups Research – CLL Advocates NZ)
It highlights the major themes and issues that emerged from the research and sets out priority areas of focus for CLLANZ. Get in touch if you’d like to help with these! Meanwhile we’re working with the facilitator of the focus groups to follow up on her recommendation that we survey a wider group of patients and carers.
Local support groups for CLL patients
Another action arising from the focus groups is the need for local support groups.
Are you interested in joining or starting one in your region?
Please contact Diane Ward: dianew564@gmail.com to find out more about this initiative.
The FixRx Buyers Club
We’re currently checking out this buyers’ club that helps patients across the world access authentic medications at affordable cost. If you have any experience of this or are interested in the concept let us know: FixRx Buyers Club
‘Fabulous ladies in the same boat’
Read this great story about three women who met online through being in the same boat - living with CLL. Despite different ages and lives, the three have supported each and become great friends while sharing and navigating the challenges of CLL: Fabulous ladies in the same boat
CLL Advocates Give a Little page
Please help support our mission. No matter how small, your donation will help us advocate for better treatments for CLL and improve the lives of those living with this disease.
Thanks to those who’ve already made donations: CLL Advocates Give a Little Page
Frequently asked questions
A recent update to the "About CLL" section on the webpage includes section that answers common questions about CLL: Frequently asked questions
It also includes a list of questions patients can ask their doctors to better understand their diagnosis and treatment options. Questions for your doctor
Please send any feedback to trustees@clladvocates.org
Best wishes
The Trustees
CLL Advocates NZ
CLL Advocates June Newsletter
24 June 2022


Our Thoughts at Matariki
Mānawatia a Matariki
We feel it’s appropriate on this special day to remember and celebrate the life of the founder of CLLANZ, Dr Neil Graham. Neil was a very generous, energetic, and compassionate human being who gave his time freely to help improve the lives of New Zealanders living with CLL. His work helped create a ‘community’ of CLL patients, through his personal engagement with patients, creation of the ongoing private CLL Advocates Facebook Group, and production of the first dedicated CLL Patient Booklet for New Zealanders. With a relentless focus on advocating for access to CLL treatments, Neil also raised the profile and understanding of CLL among politicians, Pharmac officials and other policy decision makers. He played a vital role in gaining funding for venetoclax.
Looking to the future, as is also appropriate on this day, the fight for better treatments goes on. In particular, after all these years of waiting, we’d like to see funding for ibrutinib for patients in need of it. But at this stage there’s no sign of any progress on that. While we welcome the release (finally!) of the Pharmac Review Panel report, we’re not greatly confident that it will lead to real change in the nature, speed, and transparency of Pharmac’s decision-making processes. After a three-month delay, releasing the report on the day Prime Minister Ardern met President Joe Biden suggests the Government may not have wanted it to get much attention! We have noted a change in Pharmac’s language but are keen to see some meaningful change in their processes.
We’re very pleased to say that CLLANZ Trustee Dr Gillian Corbett (see her bio Dr Gillian Corbett) has agreed to take on the role of Medical Director of CLLANZ.
You can contact her at: trustees@clladvocates.nz
Gillian will be able to lead or advise on our advocacy activities, but we are still looking for help to progress the priorities we outlined in our last newsletter. Please do get in touch with us if you can help in any way.
We hope you’re having a happy Matariki with family and friends, and we send our warmest thoughts and wishes to Neil Graham’s family and our thanks for all he did for New Zealanders living with CLL.
CLLANZ Trustees
Our Advocacy Priorities for 2022 - CLL Advocates April Newsletter
Our Advocacy Priorities for 2022
CLL Advocates April 2022 Newsletter
April 2022
Dear Friends,
The year is well under way, and we’re firmly focused on where we need to direct our energies and resources this year. We’ve agreed on the following as the key priorities for improving outcomes for CLL patients:
- Access to new treatments, notably BTK inhibitors, for relapsed CLL
- Reform of first line therapy
- Equitable access to clinical trials of unfunded CLL treatments
- The availability in NZ of testing to identify the IgVH mutation
- Prevention and management of infections in CLL patients, and
- Prevention or early detection of secondary malignancies in CLL patients.
As well as these CLL specific goals, we want to pursue the broader goal of achieving an effective, more meaningful patient voice in medicines funding decisions in NZ. (More on this in a future newsletter).
This is an ambitious agenda! And we are a very small group, but we believe with your engagement and support, and the help of our donors, we will be able to make some headway.
We have a workplan for the year, and early on in the agenda is a series of patient focus groups to identify the specific issues of most importance to you, and how we can best help you with these. We’re also keen to hear any experiences you’ve had that can help us focus our advocacy efforts effectively.
These would initially be small groups of, say, four to six, and would be on Zoom, depending on the Covid environment and geographic constraints. They would be facilitated, friendly, café style discussions. Caregivers and family members would also be welcome.
We will get in touch with you soon to set up these discussions, but in the meantime please can you get in touch with us at trustees@clladvocates.nz to let us know if you’re interested.
Help required!
We’re also still greatly in need of help to keep CLLANZ going, so if there’s any task you’re willing to take on, no matter how small, please let us know!
Best wishes
CLL Advocates NZ Trustees
CLL Advocates December Newsletter
December Newsletter - Pharmac Review Panel's interim report
Dear Friends
As you probably noted the interim report of the Pharmac Review Panel was released by Hon Andrew Little on 2 December.
It is a damning report, with the Panel noting that it could not make a meaningful analysis of Pharmac’s performance as it ‘zealously guards information’ and has a ‘fortress mentality’.
It states for example, that “We are unable to see and measure the links between inputs, impacts, outcomes, and the long-term objective of achieving the best health outcomes possible from its budget.”
It provides the following summary of Pharmac’s stakeholder engagement and an initial assessment of its decision-making processes:
- “Pharmac is underperforming in helping to remove inequitable health outcomes
- Its prioritisation approach appears to disadvantage Māori, Pacific people, disabled people and those with rare disorders
- Te Tiriti o Waitangi principles are largely unseen in decision-making processes
- There may be an excessive focus on containing costs – and a concern the cost-saving model may not be the right one to meet future health needs
- Decision making is opaque and is perceived as being slow
- Engagement with consumers and patient advocacy groups needs to be more meaningful
- Convoluted procurement processes put off pharmaceutical companies
- A perception New Zealand is falling behind other developed countries”
CLLANZ made a submission to the Panel which was very much in tune with these findings, and we were pleased to see an acknowledgement in a message in the report from the Panel’s Chair of the specific contribution made by the late Dr Neil Graham.
The final report, which will include recommendations, will be received by the Minister on 28 February, and released some time after that.
Meanwhile the Government’s Pae Ora Legislation Select Committee is considering the Pae Ora Bill that establishes the new health system structure. This bill includes Pharmac with its statutory objectives and functions quite unchanged so we have again submitted on the need to amend these and also to wait for the final report of the Review Panel before advancing this part of the legislation. See our submission here.
This has been a difficult year for CLLANZ with a very sad ending, but we are doing our best to maintain our advocacy for New Zealanders living with CLL and we look forward to continuing to do so.
We wish you the very best for a safe and peaceful Christmas and New Year.
CLL Advocates November Newsletter
November Newsletter - Neil Graham

Dear Friends of CLL Advocates NZ
We were deeply saddened to report to you last month that our Founder and Executive Director Dr Neil Graham passed away on November 15. Neil was the driving force and inspiring leader behind CLL Advocates and gave an enormous amount of his time and professional expertise to establishing the organisation and advocating for the interests of CLL patients. We miss him greatly.
As a physician and CLL patient himself, Neil gave a great deal of personal time to talking with and meeting CLL patients who had made contact through the private Facebook page, especially people who had been newly diagnosed. We know from their feedback that this was greatly appreciated. Neil also maintained a persistent and consistent focus on getting better treatments for CLL patients, regularly engaging with Pharmac officials, petitioning and making submissions to Parliament, appearing before the Health Committee and the Pharmac Review Panel and engaging with media.
Neil felt strongly about improving public understanding of CLL and providing better information for people living with it, and was able to put together a dedicated CLL patient information booklet through forging a very productive collaboration with a UK blood cancer advocacy group. In the same vein he developed a relationship with the international CLL patient group CLL Advocates Network resulting in our organisation becoming one of the first 9 members of what is now a 34 member country network sharing information and resources on CLL.
You may recall in his last (October) newsletter Neil asked for feedback on our activities and in particular any offers of help with any of our work. If you are able to volunteer any time even for small tasks, this would be greatly appreciated, especially as we move into this new era, without Neil at the helm.
You can contact us at info@clladvocates.nz.
With best wishes
CLL Advocates Trustees
Request for Feedback - October Newsletter
October Newsletter - Request for Feedback
Dear Friends,
Our monthly newsletter has now been going for a year and a half, and I’d like to hear back from you on what you think of it, and how you think we could improve it and make it as useful as possible. I’d also like to have your feedback on some of our other activities, and whether they’ve been useful to you, and your thoughts on some questions we are currently working on. In particular, I’d really appreciate an email at Neil@clladvocates.nz on all or any of the following:
Choice of topics so far, format, and topics you would like addressed in future editions. Suggestions for expanding the readership, and anything you may be able to contribute to this.
We are closing in on the “100” milestone, but there are at least 2,000 NZers with the condition, many of whom would benefit from what CLLANZ has to offer them, as would their friends and families. Please think about how you could assist with this.
Are there comments/suggestions regarding our website – content, usefulness, additional material you would like to see? How can we extend subscribers and visitors to the site?
- Research
We have another summer studentship getting underway this coming summer, a study on how many NZ CLL patients are taking ibrutinib, and questions about access, funding, response monitoring etc. This is the second we have undertaken, with funding from the Bay of Plenty Medical Research Trust. Do you have ideas on research projects we could consider for next year’s scholarship?
We would really like to expand this section. Please consider telling us yours, which you are welcome to do anonymously.
Have you read it and do you find it useful?
- Focus groups
Would you be interested in taking part in an online focus groups to discuss what would be the most useful support we could provide for you as a CLL patient or family/friend. CLL related questions?
- Clinician-led webinars/ Q&A or small group discussion sessions
Would these be of interest? I know that some informal groups that have been formed through connections on our Facebook page.
- Advocacy activities
Hopefully you’re aware of our various advocacy activities. These have included significant engagement with Ministers, Members of Parliament, the Health Select Committee, Pharmac and other relevant government health agencies, the Pharmac Review Panel, other patient advocacy groups, submissions, seminars, presentations, petitions and marches. Do you think this work is useful? Are you interested in taking part?
- Succession planning & volunteer help
Recently, two of the key CLLANZ drivers became unwell at the same time, and there was a period where CLLANZ ground to a standstill. If we had some volunteers willing to step up to temporarily covering roles at these times, that would be helpful.
On that note I’d like to record that my own health has deteriorated significantly, and I will need to pass the CLLANZ baton to someone willing and in a position to take this on. If you’re interested or have a suggestion for someone suitable, please do email me at Neil@clladvocates.nz
Thank you for your support and encouragement over the past few years.
Best wishes,
Neil Graham
CLLANZ Newsletter September 2021 - Learn the latest international developments in CLL treatments
CLLANZ Newsletter September 2021 - Learn the latest international developments in CLL treatments
Dear Friends,
I hope you’re well.
Learn the latest international developments in CLL treatments
One of the few positives that have come out of the Covid pandemic is the ability to attend high value international conferences without having to travel or pay! The international CLL Advocates Network (CLLAN) of which we are a member is holding its annual CLL Horizons Conference online on 5 – 7 November.
This will be a great multidimensional update on CLL, with contributions from leading international CLL specialists and researchers, patients and many other individuals and groups. I strongly recommend that you register for this year’s one. I attended and presented at their Edinburgh conference in 2019 and am also on a panel on this one. We’re not sure yet how many delegates we can have attend free of charge, but will clarify this and let you know.
Details of the programme and how to register, are all here on our website, and you can send any questions to info@clladvocates.net
Contribute to a global leukaemia patient experience Survey
Another collaborative project CLLAN is supporting is a global survey to help us understand the key issues, experiences and unmet needs for leukaemia patients, covering CLL, AML, ALL and CML. This is a very valuable initiative and I warmly encourage you to take the 20 minutes required to complete it.
You can find more details about it here on our website and you can access the survey here.
Meanwhile, where is the Pharmac Review Panel’s interim report??
On a final, but important note, the preliminary report of the Independent Pharmac Review Panel which was due on 20 August, is still not available over a month later. The “delay” has been attributed to lockdown (which began on August 18). This is not an adequate excuse for such an important document, which many clinicians and patient advocacy groups, including CLLANZ, went to great lengths and effort to contribute to, both in appearing before the committee and putting in very substantial submissions. For voluntary, patient-based organisations to produce and deliver this material within the very short time frame set by the Panel, was in itself a significant effort.
Somewhat ironically, Pharmac’s lack of transparency and timeliness around their processes for considering urgently needed, currently unfunded, life-saving medications, was one of the key issues raised by submitters. Failure on the part of the panel to deliver its report on time without a reasonable excuse is simply not good enough.
Best wishes
Neil Graham

Neil Graham